Intellectual Property
Protecting that spark can involve ensuring funding for early stage development of a new therapy, it can be creating the right environment for collaboration between research partners, it can be evolving the regulatory framework to keep pace with rapidly advancing science and protecting the spark means having a strong and effective intellectual property framework.
Intellectual property (IP) is the most commonly used term to describe the framework of incentives and rewards that encourage and protect innovation, driving research and development investments to areas of unmet medical need. Protecting the spark of an idea until it becomes a new treatment or cure. Pharmaceutical incentives and rewards deliver access to today’s medicines and investment into the cures of tomorrow for patients who need them.
This framework of measures such as patents, supplementary protection certificates and regulatory data protection are the foundations on which innovation is built. They provide companies researching and developing new medicines the certainty that if a medicine makes it to the market, it will be protected from unfair competition for a limited period time. This is what they need to invest in the long, complex and risky process of delivering new medicines to patients, to healthcare systems and to society and what paves the way for low-cost generics to be used by healthcare systems.
A comprehensive suite of incentives is needed to foster the development of solutions to patients’ unmet needs and turn basic science into medicines. They have enabled the research and development of today’s medicines and will lead to tomorrow’s cures.
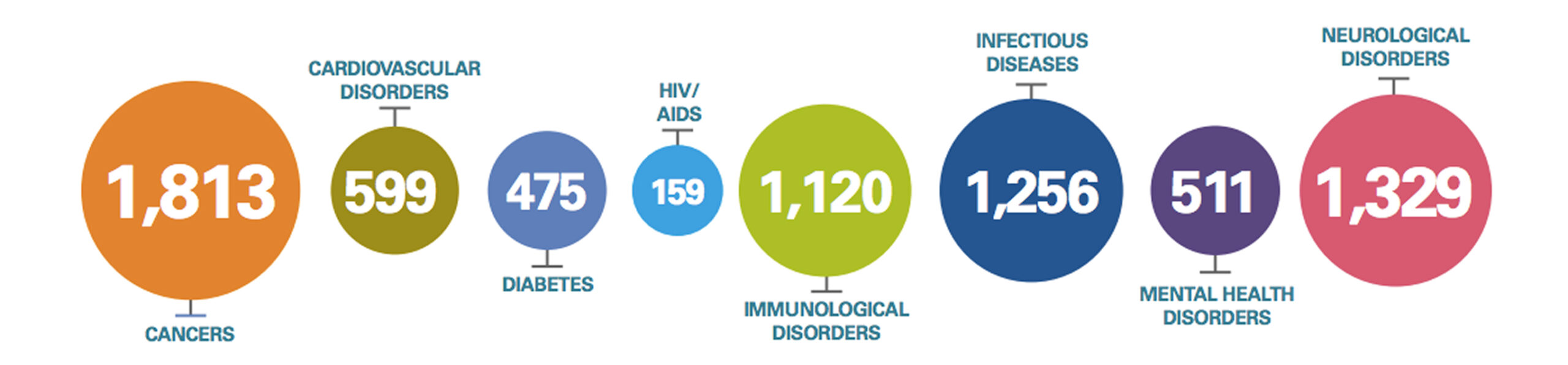
With over 7000 medicines in development, the system is working: it enables a pipeline of this scale despite the high risk of failure. These new treatments will continue to change patients’ lives; slowing disease progression, avoiding illness and reducing overall costs for healthcare systems. This exciting new wave of medical innovation and the products it will bring would simply not exist without pharmaceutical incentives and rewards.
The pharmaceutical incentives regime is a complex ecosystem that takes into account the realities of biopharmaceutical innovation and societal needs. Changing any part of the pharmaceutical incentives and rewards framework risks undermining the innovation process that patients, healthcare systems and society are relying on to address the challenges they face. The EU’s adoption of the SPC manufacturing waiver regulation is one such example, sending a negative signal that Europe is weakening its intellectual property framework, making Europe a less attractive place to invest, conduct research and impacting our ability to develop new treatments and cures for patients.
European policy makers need to find ways to redress the innovation balance, finding new ways to #ProtectTheSpark and restore global confidence in Europe as a research destination, if Europe is to realise its potential as a world leader in medical research and development.
IP-PACT(.pdf) ENG
More information please find below link:
https://www.efpia.eu/about-medicines/development-of-medicines/intellectual-propertv/
Regulatory data protection (RDP)
A patent protects inventions, RDP protects data.
In order to obtain a marketing authorisation, pharmaceutical companies need to submit extensive data relating to preclinical and clinical trials to demonstrate the quality, efficacy and safety of the medicine to be approved. Regulatory data protection (RDP) protects innovative companies’ investment in generating this extensive body of data through a limited period of exclusivity on the data, starting from marketing authorisation.
After the RDP period, generics are permitted to rely upon the originator’s data to obtain approval for their products through an “abbreviated” application. Such time limited protection is crucial to incentivize the significant investment necessary to demonstrate the safety and efficacy of new medicines, while nevertheless enabling other companies to eventually register their products on the basis of this data.